As global urbanization continues to expand, creating densely populated areas, strains on water infrastructure are expected to increase 40 percent by 2030. This is leading to a rise in the incidents of waterborne illnesses in these communities. Many existing or under development water purification systems deliver hygienic water by using UVC light in the range of 200 nm to 300 nm to effectively deactivate these pathogenic microorganisms.
Low pressure mercury lamps traditionally used in these systems have a single line emission at 254 nm. Because of the maturity of this technology, there is a plethora of research available based on microbe’s response to this exact wavelength. However, with the commercialization of UVC LEDs, an understanding of the microbe’s sensitivity to other UVC wavelengths is critical to designing effective disinfection systems.
The spectral sensitivity of a microbe is defined as the relative ability of the microbe to absorb a photon as a function of wavelength over a range of wavelengths. Figure 1 (below) shows the relative spectral sensitivity for several common pathogens referenced to a 254 nm low-pressure mercury lamp.
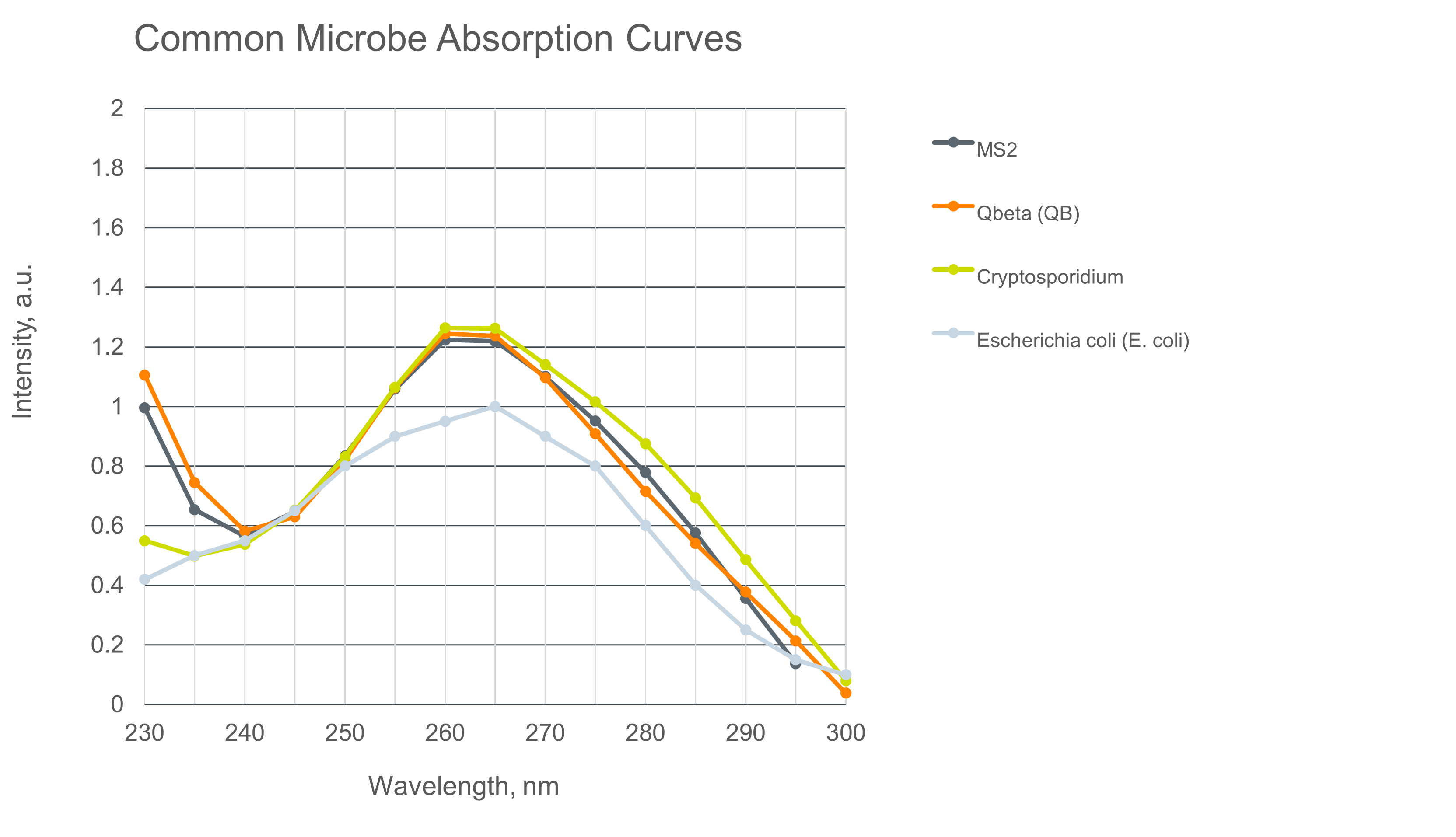
Examining these pathogens i.e. Prokaryotic (bacterial) cells E. Coli and MRSA, Eukaryotic cells (Protozoan) like “Crypto” (Cryptosporidium) and a common Obligate (virus) like Rotavirus reveals that each has a unique radiation absorption “fingerprint.” In other words, they absorb photons differently at different wavelengths based on their physical biology. While different, each pathogen demonstrates a peak absorption near 265 nm and diminishes quickly above 275 as the emission enters the UVB range. For most pathogens, there is a steep drop in sensitivity below 250 nm – for this reason, the 250 nm to 280 nm range is typically referred to as the germicidal UVC range.
The principal way UVC inactivates a pathogen is the creation of Thymine dimers within the cell DNA – which has a demonstrated peak absorption of 260 nm. However, as Figure 1 shows, the observed absorption peak and height is slightly longer and different between microbes.
So why are these absorption spectras slightly different across pathogens? This change is the result of cellular protein, specifically the presence of aromatic amino acids such as tryptophan and tyrosine which have a peak absorption close to 280 nm. Cells with more of or less of these proteins effectively screen the nucleic acids and shifts the action spectrum from 260 nm to a point between 260 nm to 270 nm, with 265 nm being the most commonly observed peak.
However, some pathogens, Rotavirus being one example, show a strong absorption peak at very short UVC wavelengths close to 230 nm. This is attributable to absorbance by peptide bonds which surround the RNA. Since this attribute is limited to a narrow set of pathogens most practical UV-C disinfection products focus on emitting in the germicidal UV-C range to ensure consistent disinfection across all pathogens.
Given these differences, what should an engineer do when designing UVC disinfection systems? The best approach is to understand the spectral sensitivity of your target microbe and ensure that you have adjusted your required UV dose to the wavelength that you’re using. However, this is not always feasible or plausible, especially when designing to a cocktail of target microbes. In these cases, selecting a UV source that provides significant emission in the key germicidal wavelengths from 260 nm to 270 nm ensures the most predictable disinfection performance.